
Yes, silver tarnishing is a chemical change because it involves a reaction between the metal and atmospheric sulfur that creates a new substance called silver sulfide. For jewelry retailers and wholesalers, maintaining the pristine appearance of inventory is a constant challenge as metals naturally lose their luster over time.
When your customers notice a dark, dull film on a piece of jewelry, they immediately question the quality and purity of the metal. The underlying concern is whether is silver tarnishing a chemical change that permanently damages the item or simply a surface-level issue that requires basic cleaning. Misunderstanding the science behind this process often leads to inadequate storage solutions and significant losses in stock value.
By exploring the molecular shifts in precious metals, you can implement superior preservation strategies for your collection. Partnering with a specialist like YiXin Jewelry Packaging allows you to utilize advanced materials that block reactive elements before they reach your products.
Is silver tarnishing a chemical change in jewelry?

Silver tarnishing is a chemical change because it results in the formation of a new chemical compound on the surface of the jewelry. When managing high-volume retail stock, you must understand that the darkening of silver is not just a physical stain. If you are currently using standard custom jewelry boxes , you need to evaluate if your materials are effectively preventing this specific chemical shift.
What defines a chemical change in precious metals?
A chemical change occurs when the original substance transforms into a different substance with unique properties. In the case of jewelry, the silver atoms bond with sulfur atoms to create a dark layer that did not exist previously.
Think about this:
- The formation of silver sulfide (Ag₂S) is the primary indicator of tarnish.
- This change is irreversible without using a chemical reduction or abrasive polishing.
- The surface properties, such as conductivity and reflectivity, are fundamentally altered.
How does tarnish differ from surface dirt or grime?
Surface dirt is a physical accumulation of external particles that can be wiped away without affecting the metal’s structure. Tarnish, conversely, is an integrated layer of the metal itself that has undergone a molecular transformation.
Key Takeaway: Tarnish is not a simple cleaning issue; it is a structural transformation of the metal’s surface into a new compound.
| Change Type | Outcome | Reversibility | |
|---|---|---|---|
| Physical (Dirt) | Surface accumulation | Easy (Wipe) | |
| Chemical (Tarnish) | New compound formation | Hard (Chemical/Abrasive) |
This comparison highlights why your storage must focus on preventing chemical reactions rather than just keeping items clean.
Why is silver tarnishing a chemical change for metals?

Metals experience a chemical change during the tarnishing process because the atoms of the metal share electrons with environmental reagents to create a stable compound. The primary question of why is silver tarnishing a chemical change is answered by the presence of sulfur in the air. This reaction creates a thin, protective yet unsightly film that covers the original shiny metal.
What are the primary reactants in the tarnishing process?
The reaction requires specific elements to proceed, most notably hydrogen sulfide gas which is often found in urban environments. Even in trace amounts, these sulfur compounds are aggressive enough to start the bonding process immediately upon contact.
Here is the kicker:
- Hydrogen sulfide (H₂S) acts as the primary reactant in most jewelry stores.
- Atmospheric moisture often serves as a catalyst to speed up the bonding.
- Oxygen, while a factor for some metals, is less reactive with pure silver than sulfur.
How does the formation of silver sulfide occur?
When silver atoms meet sulfur molecules, they undergo an ionic reaction that produces silver sulfide. This new substance is black or dark grey, which explains why your bright silver pieces eventually turn dark.
Key Takeaway: The creation of Ag₂S represents a distinct chemical transition from a pure element to a metal sulfide compound.
| Reactant A | Reactant B | Resulting Compound | |
|---|---|---|---|
| Pure Silver (Ag) | Hydrogen Sulfide (H₂S) | Silver Sulfide (Ag₂S) |
This chemical equation demonstrates that the metal is physically and chemically changing its identity at the surface level.
Is silver tarnishing a chemical change or physical?

Tarnishing is classified as a chemical change because it involves the breaking and forming of chemical bonds to create a new substance. Wholesalers often transition to PE film jewelry boxes to create airtight barriers once they realize that is silver tarnishing a chemical change driven by air exposure. Understanding this distinction is vital for choosing packaging that prevents atmospheric interactions.
Why isn’t tarnish considered a physical alteration?
A physical change only alters the form or appearance of a substance, like melting silver or bending a wire. Because the silver is actually bonding with sulfur to become a sulfide, it no longer remains just silver, which confirms the chemical nature.
But wait, there’s more:
- Bending or scratching silver does not create a new substance.
- Tarnishing changes the chemical formula of the surface layer.
- The process releases hydrogen gas as a byproduct of the sulfide formation.
What properties change during the tarnishing process?
During this transition, the metal loses its characteristic luster and takes on a dull, brittle texture. This degradation of the surface can eventually lead to pitting if the chemical reaction is allowed to penetrate deeply into the alloy.
Key Takeaway: Physical changes preserve the identity of the substance, whereas tarnishing creates an entirely new chemical entity.
| Feature | Physical Change (Bending) | Chemical Change (Tarnish) | |
|---|---|---|---|
| New Substance | No | Yes (Silver Sulfide) | |
| Color Change | No | Yes (Yellow to Black) |
Identifying these differences helps you explain to clients why professional storage is a necessity rather than an option.
How is silver tarnishing a chemical change identified?
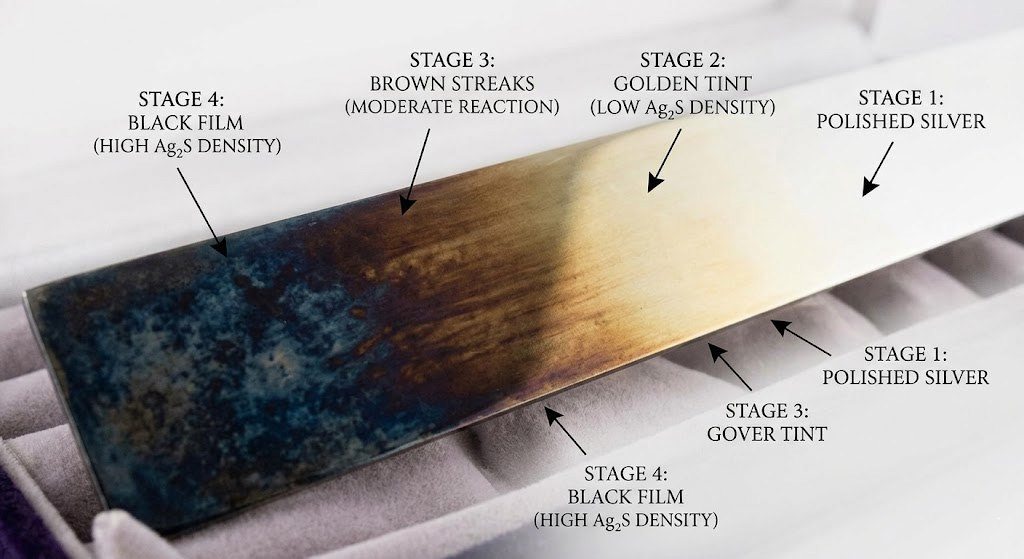
You can identify that silver tarnishing is a chemical change through the observation of progressive color shifts and surface dullness that cannot be removed with water alone. Monitoring your inventory for these signs allows you to intervene before the reaction affects the entire piece. Understanding how is silver tarnishing a chemical change identified ensures your staff can spot early-stage degradation.
What visual cues indicate a chemical reaction is occurring?
The first sign of tarnish is often a slight yellowish or golden tint on the silver surface. As the chemical reaction continues and the layer of silver sulfide thickens, the color deepens into brown and eventually a solid, matte black.
You might be wondering:
- Why does the color change so predictably?
- The thickness of the sulfide layer determines which light wavelengths are reflected.
- Early stages reflect yellow, while thick layers absorb most light, appearing black.
Can you differentiate tarnish from simple surface wear?
Surface wear usually presents as small scratches or a loss of polish due to friction, which are purely physical marks. Tarnish appears as a uniform or mottled film that covers the metal regardless of whether the item has been handled or moved.
Key Takeaway: The specific progression from yellow to black is a hallmark of the silver-sulfur chemical reaction.
| Stage | Visual Appearance | Chemical Intensity | |
|---|---|---|---|
| Initial | Light Yellow Tint | Low Sulfide Density | |
| Intermediate | Brown/Blue Streaks | Moderate Reaction | |
| Advanced | Deep Black Film | High Sulfide Density |
Using this visual guide helps you categorize the health of your inventory and schedule necessary restoration.
Is silver tarnishing a chemical change from oxidation?

While many people use the terms interchangeably, silver tarnishing is a chemical change primarily caused by sulfidation rather than pure oxidation. For high-end inventory, using specialized necklace boxes can help mitigate the specific elements that cause these reactions. Knowing that is silver tarnishing a chemical change involving sulfur helps you select packaging that targets the correct atmospheric threats.
Does oxygen play a role in sterling silver tarnish?
Pure silver does not oxidize easily in normal temperatures, but sterling silver contains copper which is highly reactive with oxygen. In this case, the copper within the alloy undergoes oxidation, contributing to the overall darkening of the jewelry piece.
It gets even more interesting:
- Sterling silver is 7.5% copper, making it more prone to oxidation than 999 silver.
- Oxygen and moisture work together to accelerate the corrosion of base metals in alloys.
- A combination of copper oxide and silver sulfide creates a very stubborn tarnish.
What environmental catalysts speed up this reaction?
Humidity is the most common catalyst in retail environments, as moisture on the metal surface provides a medium for ions to move. Additionally, atmospheric pollutants and even the oils from human skin can provide the reactants needed to kickstart the chemical process.
- High humidity levels in the showroom.
- Airborne pollutants from nearby industrial activity.
- Salts and acids from frequent customer handling.
Key Takeaway: While sulfur is the main culprit for silver, oxidation of copper within the alloy significantly accelerates the tarnishing process.
| Catalyst | Source | Effect on Reaction | |
|---|---|---|---|
| Humidity | Air/Weather | Speeds up ion exchange | |
| Copper Content | Alloy Mixture | Increases oxidation rate |
Managing these environmental variables is the second most important step after selecting the right protective packaging.
Is silver tarnishing a chemical change preventable?
Silver tarnishing is a chemical change that is highly preventable through the use of barrier protection and chemical neutralizers. As a B2B professional, your goal is to stop the chemical clock before the metal surface has a chance to react with the air. Because is silver tarnishing a chemical change that depends on external gases, removing those gases from the equation is the most effective solution.
How does airtight storage stop chemical reactions?
Airtight storage works by physically separating the silver atoms from the sulfur molecules present in the atmosphere. Without the presence of a reactant (sulfur), the chemical reaction simply cannot occur, regardless of how much time passes.
Let’s face it:
- Standard open displays are the most high-risk environments for silver.
- Vacuum sealing or airtight containers provide the highest level of protection.
- Reducing the volume of air around the jewelry limits the available sulfur.
Which packaging materials help neutralize sulfur?
Advanced packaging solutions often incorporate anti-tarnish strips or specialized coatings that actively absorb sulfur. These materials act as a “sacrificial” surface, reacting with the sulfur before it can reach your valuable jewelry items.
- Anti-tarnish tabs or strips placed inside boxes.
- Sulfur-free tissue paper for wrapping individual pieces.
- High-quality plastics that do not off-gas harmful chemicals.
Key Takeaway: Prevention relies on a dual strategy of physical isolation and active chemical neutralization within the storage environment.
| Method | Type of Protection | Best Use Case | |
|---|---|---|---|
| PE Film Boxes | Physical Barrier | Long-term inventory storage | |
| Anti-Tarnish Strips | Chemical Absorption | Point-of-sale packaging |
Implementing these methods ensures that your stock remains in “showroom ready” condition for much longer periods.
Is silver tarnishing a chemical change for all alloys?
Whether silver tarnishing is a chemical change for a specific item depends largely on the alloy’s composition and the presence of reactive metals. When selecting bracelet boxes , you must consider how the acidity of the material might interact with different metal purities. Not all jewelry responds identically when you ask if is silver tarnishing a chemical change because gold and platinum have different reactivity levels.
Why do different gold karats tarnish at different rates?
High-karat gold, like 18k or 24k, is extremely stable and rarely undergoes chemical changes under normal conditions. However, 10k or 14k gold contains higher percentages of silver and copper, which are the elements that actually react with sulfur and oxygen.
Surprisingly enough:
- 24k gold is chemically inert and does not tarnish.
- 10k gold can tarnish significantly due to its high base metal content.
- The “gold” tarnish you see is usually the silver in the alloy turning into silver sulfide.
Does copper content increase the likelihood of tarnish?
Copper is highly susceptible to both oxidation and sulfidation, making it a major driver of tarnish in sterling silver and low-karat gold. If your alloy has a high copper concentration, the chemical transformation will happen much faster and produce a darker, more greenish residue.
- Copper reacts with oxygen to form brownish-black copper oxide.
- Copper reacts with sulfur to form black copper sulfide.
- Higher copper percentages lead to faster overall degradation of the finish.
Key Takeaway: The chemical stability of a jewelry piece is directly proportional to its noble metal content and inversely proportional to its base metal content.
| Metal/Alloy | Reactivity Level | Primary Reaction | |
|---|---|---|---|
| Sterling Silver | High | Sulfidation | |
| 14k Gold | Moderate | Oxidation/Sulfidation | |
| 24k Gold | Zero | None |
This ranking allows you to prioritize which items in your collection require the most rigorous protective packaging.
Is silver tarnishing a chemical change that can be reversed?
Silver tarnishing is a chemical change that can be reversed by using another chemical reaction to break the bonds of the silver sulfide. When your inventory has already darkened, you must choose between mechanical removal or chemical restoration to bring back the shine. Knowing how is silver tarnishing a chemical change reversed helps you save stock that would otherwise be unsellable.
How do chemical dips restore the metal’s surface?
Chemical dips contain reducing agents that react with the sulfur in the silver sulfide, stripping it away and leaving the original silver atoms behind. This is a highly efficient way to restore complex designs that are difficult to reach with a polishing cloth.
Here’s the deal:
- Dips use a chemical process to “undo” the sulfide bond.
- This method is fast and requires very little labor for high-volume retailers.
- Overuse can eventually dull the finish by stripping away a microscopic layer of metal.
Is physical polishing enough to remove silver sulfide?
Physical polishing uses mild abrasives to mechanically “scrape off” the layer of silver sulfide from the surface. While effective, this is technically a physical removal of the chemically changed layer rather than a reversal of the reaction itself.
- Polishing cloths are best for lightly tarnished items.
- Abrasive pastes can remove deep tarnish but may change the item’s texture.
- Physical polishing is time-consuming for large wholesale inventories.
Key Takeaway: True reversal happens through chemical reduction, while polishing physically removes the damaged layer of metal.
| Method | Action | Best For | |
|---|---|---|---|
| Chemical Dip | Bond Breaking | Intricate designs/bulk | |
| Polishing | Abrasive Removal | Flat surfaces/light tarnish |
Choosing the right restoration method depends on the value of the piece and the severity of the chemical transformation.
Is silver tarnishing a chemical change caused by sulfur?
The most frequent reason silver tarnishing is a chemical change is the introduction of sulfur from low-quality packaging materials. Investing in premium ring boxes ensures that the is silver tarnishing a chemical change doesn’t become a recurring problem caused by your own display items. You must be vigilant about the “invisible” sulfur sources that may be lurking in your storage room.
Where do sulfur compounds in retail environments come from?
Sulfur can be found in surprising places, including rubber bands, certain types of paper, and even the cardboard used in shipping. When these materials are kept in close proximity to jewelry, they “off-gas” sulfur compounds that settle on the metal and begin the chemical reaction.
The bottom line?
- Common rubber bands are a major source of concentrated sulfur.
- Unrefined paper products often contain sulfur used in the bleaching process.
- Wool and silk fabrics naturally contain sulfur proteins that can react with silver.
Can low-quality packaging accelerate the tarnishing process?
Yes, cheap adhesives and foams used in budget jewelry boxes often release volatile organic compounds that act as reactants. Instead of protecting your jewelry, poor packaging can actually create a “gas chamber” effect that turns your silver black within weeks.
- Glues used in box construction may contain sulfur-based curing agents.
- Low-grade foams can break down and release corrosive gases over time.
- Non-archival quality linings may trap moisture against the metal.
Key Takeaway: Your packaging choice is either a shield against chemical change or a catalyst for it; there is no middle ground.
| Material | Safety Status | Risk Level | |
|---|---|---|---|
| Acid-Free Paper | Safe | Low | |
| Rubber/Elastic | Dangerous | High | |
| Specialized PE Film | Recommended | Minimal |
By selecting laboratory-tested, sulfur-free materials, you eliminate the most common cause of chemical degradation in jewelry.
FAQs: Is silver tarnishing a chemical change or not?
Can I stop silver from tarnishing forever?No, it is technically impossible to stop the chemistry entirely unless you store the silver in a complete vacuum. However, using high-quality barriers and anti-tarnish technology can delay the process for many years.
How does moisture affect the rate of tarnish?Moisture acts as a powerful catalyst that speeds up the chemical reaction between silver and sulfur. High humidity allows the sulfur ions to migrate more easily to the metal surface, causing tarnish to form much faster than in dry conditions.
Is it safe to use ultrasonic cleaners on tarnish?No, ultrasonic cleaners are designed to remove physical dirt and grease using sound waves, not chemical tarnish. To remove silver sulfide, you must use a chemical restoration method or an abrasive polishing technique.
Why does my skin make silver jewelry turn black?Your skin produces sweat and oils that contain amino acids and sulfur compounds, which react directly with the silver. High acidity in an individual’s skin chemistry can cause a rapid chemical change, turning the jewelry black shortly after it is worn.
Does high-quality packaging really prevent tarnish?Yes, high-quality packaging provides a physical barrier and often contains chemical scavengers that neutralize sulfur before it can reach the metal. This intervention is the most effective way to prevent the chemical change from occurring during long-term storage.
Conclusion
Understanding the science of metal corrosion is the first step in preserving your brand’s reputation and the value of your inventory. Because is silver tarnishing a chemical change, it requires more than just a surface cleaning—it requires a proactive strategy of prevention and proper storage that addresses the root chemical causes.
Protect your inventory from the damaging effects of tarnish with premium, laboratory-tested packaging solutions. To learn more about how our custom boxes can safeguard your collections and enhance your presentation, contact us today.
